Drug pricing pressure in the United States is intensifying, and more patients are feeling the burden of affordability of essential, lifesaving medications. Approximately one in three Americans skips filling a prescription due to cost. Behind that number is a system under fire—from patients, policymakers, and pharma alike. The Trump administration has publicly committed to lowering prices for American citizens. On July 31, 2025, President Donald Trump posted on Truth Social, pressuring 17 pharmaceutical CEOs a 60 day deadline (September 29, 2025) to slash U.S. drug prices; notably urging them to match prices charged overseas.
“If you refuse to step up, we will deploy every tool in our arsenal to protect American families from continued abusive drug-pricing practices.” - President Trump on Truth Social, July 31, 2025.
While lowering health care costs and increasing access to treatment is a bipartisan goal, how these objectives are achieved is far more complex. On average, Americans pay 2.78 times more for medications than those in 33 comparable countries. Given that Americans are the largest consumers of pharmaceuticals worldwide, the burden of treatment costs is inescapable, touching the lives of millions.
Historical Context
In the 1980s, the pharmaceutical industry experienced a transformative surge, with breakthroughs in treatments for mental illness, heart disease, and cancer. This wave of innovation expanded both private and public insurance coverage, driving up demand, spending, and the use of prescription drugs and physician services.
As the clinical healthcare sector grew, the insurance landscape shifted toward corporatization, leading to steep increases in premiums. Insurance companies became more aggressive in limiting claims, leaving patients with higher out-of-pocket costs. Over time, this evolved into today’s intricate network of pharmaceutical companies, pharmacy benefit managers (PBMs), and insurers, leaving many Americans uncertain who is truly responsible for rising costs.
Global Drug Pricing Overview
The United States is a global outlier in per capita pharmaceutical spending and overall drug consumption. U.S. citizens face a stark imbalance in drug costs compared to the rest of the world, leaving many frustrated. To understand why, it is essential to examine how pharmaceutical companies set drug prices globally and how healthcare systems in peer nations manage access and affordability.
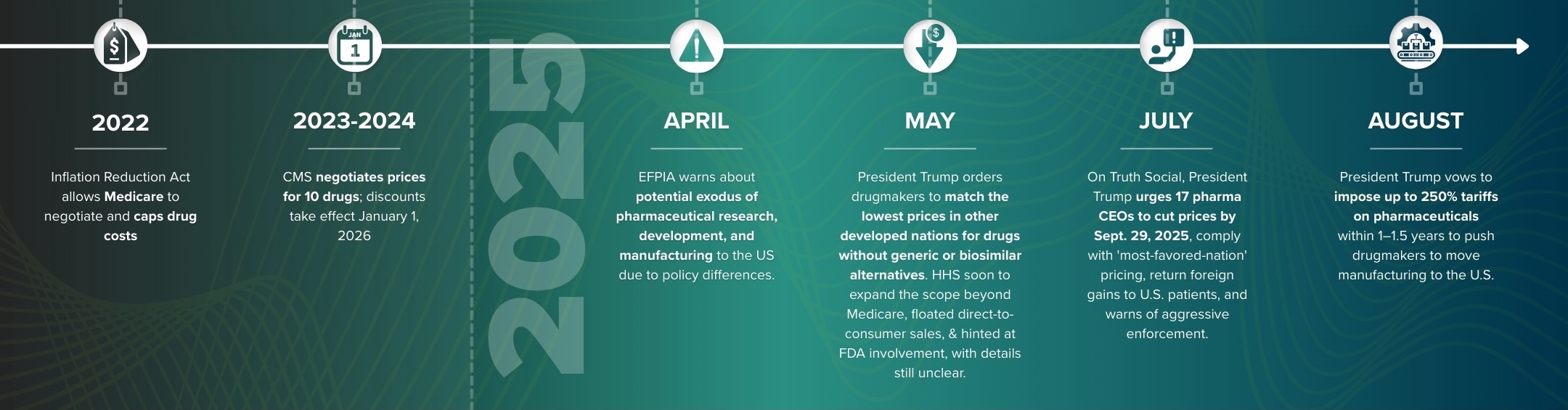
Unlike similar developed countries, the United States does not regulate drug prices because of a longstanding policy framework shaped by market-oriented healthcare principles and powerful industry lobbying. Until the Inflation Reduction Act (IRA) was implemented in 2022, Medicare was prohibited by law from negotiating directly with drug manufacturers, a restriction rooted in the 2003 Medicare Modernization Act. Policymakers argued that competition rather than government intervention would drive innovation and lower costs. In practice, this hands-off approach has allowed pharmaceutical companies to set launch prices freely and raise them over time.
Insurers and PBMs also play a significant role in driving up what patients ultimately pay. PBMs negotiate drug prices and rebates with manufacturers on behalf of insurers, but these negotiated savings are often not passed directly to patients at the pharmacy counter. Instead, rebates can be kept by PBMs or used to offset overall plan costs, which can still leave high out-of-pocket expenses for individuals. A recent study reveals that PBM rebates represented 9% of the average total healthcare cost for an employee with employer-sponsored insurance. Notably, none of those rebate dollars were used to reduce patients’ out-of-pocket prescription costs; instead, they largely went toward lowering premiums. Insurers may also structure benefit designs with high deductibles, coinsurance, or tiered formularies that place the most expensive drugs in higher cost-sharing categories. These practices mean that even when the “net price” of a drug falls due to rebates or discounts, the price paid by patients can remain high or even increase.
New Drug Pricing Order Shakes Global Pharma
On April 8, 2025, the European Federation of Pharmaceutical Industries and Associations (EFPIA) issued a statement warning that the United States now outperforms Europe on several investor metrics, including access to capital, intellectual property protections, speed of regulatory approval, and financial incentives for innovation. The group cautioned that uncertainty over tariffs offers little motivation for investment in the European Union, while the US presents a more stable and rewarding environment. As a result, some European pharmaceutical companies are considering moving manufacturing operations to the United States.
One month later, on May 12, 2025, the Trump Administration issued an executive order requiring drug manufacturers to match the lowest prices available in other developed countries, aiming to reduce US drug costs. On May 20, the Department of Health and Human Services (HHS) followed with guidance mandating price matching for brand-name drugs that lack generic or biosimilar competition. This order expands on a 2020 “Most-Favored-Nation” (MFN) policy, which had tied Medicare drug payments to the lowest international prices before that earlier rule was blocked in court and withdrawn.
The 2025 order applies beyond Medicare, potentially extending to Medicaid and even private markets. It also floats new measures such as direct-to-consumer drug purchasing and possible FDA actions, though key details remain vague. Overall, it signals a renewed and more ambitious push for global reference pricing.
Just over two months after the most-favored-nation order, 250% tariff threats, and calls to increase manufacturing in the US, President Trump used Truth Social to address 17 major pharmaceutical chief executives. In his message, he demanded steep price cuts and warned, “If you refuse to step up, we will deploy every tool in our arsenal to protect Americans from abusive drug pricing practices”. He also outlined the main goals of the most-favored-nation policy in a letter that set a 60-day deadline for action. The companies were instructed to extend most-favored-nation pricing to Medicaid, guarantee that newly launched drugs would be offered at most-favored-nation prices, return any increased revenues from foreign markets to American patients and taxpayers, and provide for direct purchasing at most-favored-nation pricing. Trump further emphasized that making higher drug prices in Europe a top priority was central to his strategy. Despite these requests, analysts, lobbyists, and drug pricing experts generally agree that the likelihood of full industry compliance remains low.
In addition to pricing mandates, the Trump Administration has taken steps to strengthen domestic pharmaceutical supply chains. On August 15th, 2025, The White House directed HHS to identify roughly 26 medicines critical to US public health and stockpile a six-month supply of their active pharmaceutical ingredients (APIs). This initiative updates a 2022 list of 86 essential medicines and aims to reduce reliance on foreign sources, particularly China, while encouraging domestic manufacturing. Building on the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR) established during the COVID-19 pandemic, the plan calls for the existing repository to begin receiving APIs within 120 days, with a second facility scheduled for completion a year later. Major pharmaceutical companies, including AbbVie, AstraZeneca, and Johnson & Johnson, have pledged investments to expand US production, reflecting a broader push to secure supply chains and reinforce US self-sufficiency.
Pharma Pushes Back: Shifting the Narrative
In response to President Trump’s renewed 2025 push to slash drug prices in America, a policy stance that mirrors his earlier attempts to rein in pharmaceutical costs, industry leaders have been quick to reframe the conversation. Rather than focusing solely on list prices, companies such as Johnson & Johnson are urging policymakers to consider the broader complexities of the drug pricing ecosystem.
In its recent Transparency Report, Johnson & Johnson disclosed that it returned nearly 48 billion dollars, representing 58% of its gross sales, in the form of rebates, discounts, and fees. Most of these funds went to intermediaries such as pharmacy benefit managers and insurers. Despite an 18.2% drop in Johnson & Johnson’s net prices since 2016, patients have seen little relief at the pharmacy counter. Rising out-of-pocket costs, the company argues, are largely the result of insurance design flaws and administrative barriers such as step therapy and prior authorizations. Johnson & Johnson’s central message to policymakers is clear: lowering list prices will not meaningfully help patients unless reforms ensure that savings are passed directly to consumers at the point of sale.
Other pharmaceutical executives have voiced similar concerns. The chief executive officer of Eli Lilly has warned that the United States is in urgent need of comprehensive healthcare reform, not simply headline-grabbing price mandates. He cautioned that if the administration moves forward with a most-favored-nation approach, which would tie United States drug prices to the lowest available internationally, it could replicate the challenges facing Europe’s pharmaceutical sector. Those markets often experience lower productivity in drug development and, paradoxically, higher costs for the United States in the long run.
Some companies are pairing their pushback with offers of compromise. AstraZeneca’s CEO announced that the company has proposed cutting prices on certain U.S. drugs, though he did not specify which ones. He emphasized the need for greater pricing transparency and expressed a willingness to adjust strategies. The move comes as AstraZeneca posts strong earnings and commits 50 billion dollars to U.S. manufacturing and research by 2030, aiming for half of its projected 80 billion dollars in revenue to come from the U.S. market.
Swiss drugmaker Roche has also indicated that it is exploring direct-to-consumer sales of prescription medicines in the United States as part of ongoing discussions with the federal government. The company says such a move could help lower costs for patients while bypassing some of the inefficiencies in the current supply chain.
Together, these responses reveal an industry determined to shift the conversation away from aggressive price controls and toward a broader discussion about how drugs are paid for, how savings are shared, and how the United States can balance affordability with continued investment in innovation.
The Road Ahead
Tackling America’s high drug prices will not come from a single policy fix. It will require a broad and coordinated effort. That begins with pulling back the curtain on the pharmaceutical supply chain so patients can finally see how prices are set and where their money actually goes. Rebates and discounts, often buried in layers of complexity, should be redesigned to deliver real savings directly at the pharmacy counter.
Some experts point to value-based pricing, which ties a drug’s cost to its actual clinical effectiveness, as one way to make pricing fairer. Others believe we cannot ignore the role of PBMs, whose opaque business practices may need closer scrutiny to ensure they serve patients rather than profit margins.
At the same time, more Americans are choosing to bypass traditional channels altogether. Services such as Mark Cuban’s Cost Plus Drugs offer a simpler model with transparent markups, no hidden fees, and prices that remain consistent regardless of who is paying. The growing popularity of these options reflects a clear demand for more straightforward and affordable access to care.
If future policies can lower out-of-pocket costs, streamline the path from prescription to patient, and put people rather than profits at the center, the United States could begin to build a drug pricing system that is transparent, equitable, and sustainable for the long term.
Read More
For more information about recent changes in policy and reaction in the healthcare industry check our blog “Drug Pricing in an Era of Tariffs” for a deep dive on tariffs and their impact on the U.S and global healthcare market access landscape.
At PFG MedComm, we continuously explore innovative approaches and can help you navigate the complexities of the healthcare payor landscape. Click here to download PFG MedComm’s Ultimate Guide to Market Access.
Need assistance in crafting revolutionized enhancements to your value communication strategy with market access stakeholders? At PFG MedComm, we continuously explore innovative approaches.
Schedule a brief intro to see how we can help!
Let's Talk